Utente:Grasso Luigi/sanbox1/Ossidazione di Boyland–Sims
Vai alla navigazione
Vai alla ricerca

L'ossidazione di Boyland-Sims 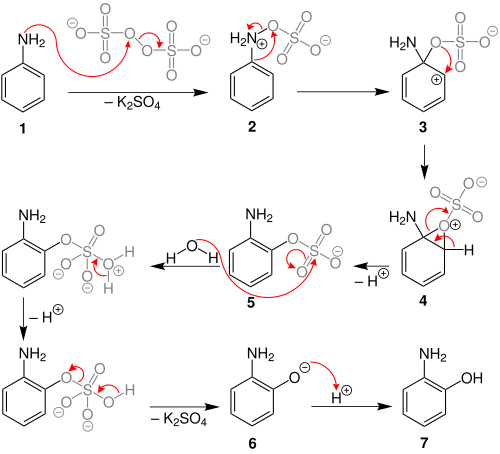
Meccanismo dell'ossidazione di Boyland-Sims
L' ossidazione di Boyland–Sims è la reazione chimica of anilines with alkaline potassium persulfate, which after hydrolysis forms ortho-hydroxyl anilines.[1][2][3]

The ortho-isomer is formed predominantly. However, the para-sulfate is formed in small amounts with certain anilines.[4]
Meccanismo della reazione
[modifica | modifica wikitesto]Behrman has shown that the first intermediate in the Boyland–Sims oxidation is the formation of an arylhydroxylamine-O-sulfate (2).[5] Rearrangement of this zwitterionic intermediate forms the ortho- sulfate (5), which then hydrolyses to form the ortho-hydroxyl aniline.

Voci correlate
[modifica | modifica wikitesto]Note
[modifica | modifica wikitesto]- ^ (EN) Boyland E.; Manson D.; Sims Peter, 729. The preparation of o-aminophenyl sulphates, in J. Chem. Soc., 1953, pp. 3623-3628, DOI:10.1039/jr9530003623.
- ^ (EN) Boyland E.; Sims Peter, The oxidation of some aromatic amines with persulphate, in J. Chem. Soc., 1954, pp. 980-985, DOI:10.1039/jr9540000980.
- ^ (EN) Behrman, E. J., The Persulfate Oxidation of Phenols and Arylamines (The Elbs and the Boyland-Sims Oxidations), in Org. React., vol. 35, 1988, pp. 421-515, DOI:10.1002/0471264180.or035.02, ISBN 0471264180.
- ^ (EN) Boyland, E.; Sims, P.; Williams, D. C., The oxidation of tryptophan and some related compounds with persulphate, in Biochem. J., vol. 62, n. 4, 1956, pp. 546-550, PMC 1215958, PMID 13315210.
- ^ (EN) Behrman, E. J., The ortho-para ratio and the intermediate in the persulfate oxidation of aromatic amines (the Boyland-Sims oxidation), in J. Org. Chem., vol. 57, n. 8, 1992, pp. 2266, DOI:10.1021/jo00034a016.
Bibliografia
[modifica | modifica wikitesto]- (EN) Behrman Edward J., On the Mechanism of the Boyland-Sims Oxidation, in Prog. React. Kinet. Mec., vol. 39, n. 3, Science Reviews 2000 Ltd, 2014, pp. 308–310, DOI:10.3184/146867814X14062204626705.